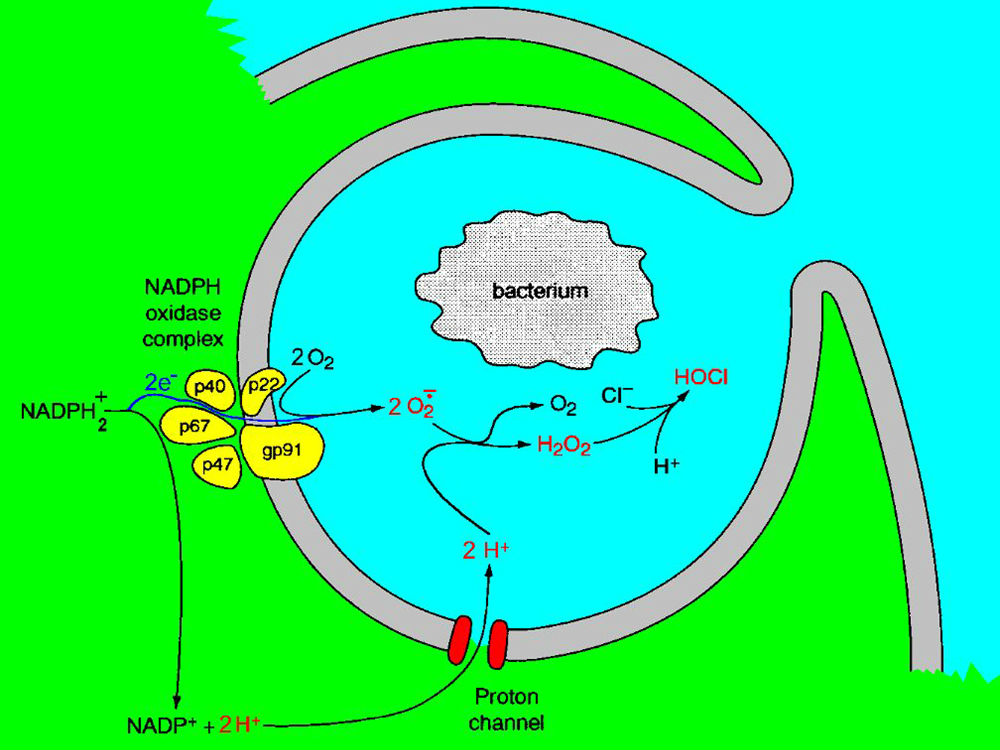
The properties and biological functions of ion channels are long-term interests of Tom DeCoursey, PhD. A major focus of his laboratory in recent years is the voltage-gated proton channel. Modulation of the voltage-dependence of this channel by pHo and pHi ensures that it opens only when the electrochemical gradient for H+ is outward (in most species). In other words, when the proton channel opens, it extrudes acid from cells.In a long collaboration with Vladimir V. Cherny, PhD, and others, they have explored the behavior of proton channels in alveolar epithelial cells and in white blood cells (human neutrophils and eosinophils). Immune cells engulf (phagocytose) bacteria and kill parasites by secreting reactive oxygen species (e.g., ChloroxTM). The enzyme responsible for these heroic actions is NADPH oxidase. This enzyme moves electrons across the cell membrane to form superoxide anion near the invading critters.
The lab measures the electron movement directly as an electrical current. For each electron, that leaves, one proton stays in the cell. To prevent massive depolarization as well as acidification, protons exit the cell through proton channels. Without H+ efflux, the killing process would be interrupted prematurely. Fortunately, proton channels are activated, relieving the cell of excess acid and preventing depolarization. The discovery of proton channels has been a great boon to cells that, until this time, had to use other, less efficient means of extruding acid
Identification of proton channel genes in 2006 has transformed the field. More functions are described each year, and structure-function studies are underway. The channel was shown to be a dimer, with a distinct conduction pathway in each protomer. The dimer gates cooperatively—both protomers need to move before either conducts. New genes (nine confirmed, dozens more speculated) are appearing at a high rate.
The proton channel resists efforts to crystallize it. The first crystal structure (which was reported by K. Takeshita and colleagues in a 2014 article) is likely closed, and is of a chimera with a voltage-sensing phosphatase.
James Letts' valiant efforts to determine the open structure of HV1 can be found in his PhD dissertation, which also nicely shows that Asp112 mutants have severely compromised H+ flux. HV1 triggers the flash in bioluminescent dinoflagellates (Smith et al, 2011).
Publications
Select recent works
(If you are unable to download any pdfs, please email Thomas E. DeCoursey, PhD)
DeCoursey, T.E. (2015). “Commentary: Structural Revelations of the Human Voltage-Gated Proton Channel.” Proceedings of the National Academy of Sciences, USA. 122:13430-13431.
V.V. Cherny, D. Morgan, B. Musset, G. Chaves, S.M.E. Smith, and T.E. DeCoursey. (2015). “Tryptophan 207 Is Crucial to the Unique Properties of the Human Voltage Gated Proton Channel, hHV1.” Journal of General Physiology. 146:343-356.
DeCoursey, T.E. (2015). “The Voltage-Gated Proton Channel: A Riddle, Wrapped in a Mystery, Inside an Enigma.” Biochemistry. 54:3250-3268.
Dudev, T.B., Musset, D. Morgan, V.V. Cherny, S.M.E. Smith, K. Mazmanian, T.E. DeCoursey, and C. Lim. (2015). “Selectivity Mechanism of the Voltage-Gated Proton Channel, HV1.” Scientific Reports. 5:10320.
Hondares, E., M.A. Brown, B. Musset, D. Morgan, V.V. Cherny, C. Taubert, M.K. Bhamrah, D. Coe, F. Marelli-Berg, J.G. Gribben, M.J.S. Dyer, T.E. DeCoursey, and M. Capasso. (2014). “Enhanced Activation of an Amino-Terminally Truncated Isoform of the Voltage-Gated Proton Channel HVCN1 Enriched in Malignant B Cells.” Proceedings of the National Academy of Sciences, USA. 111:18078-18083.
DeCoursey, T.E., and J. Hosler. (2014). “Philosophy of Voltage-Gated Proton Channels.” Journal of the Royal Society Interface. 11: 20130799.
Morgan, D.B., Musset, K. Kulleperuma, S.M.E. Smith, S. Rajan, V.V. Cherny, R. Pomès, T.E. DeCoursey. (2013). “Peregrination of the Selectivity Filter Delineates the Pore of the Human Voltage Gated Proton Channel hHV1.” Journal of General Physiology. 142:625-640. Here is the video summary.
DeCoursey, T.E. (2013). Voltage gated proton channels: molecular biology, physiology and pathophysiology of the Hv family. Physiological Reviews. 93:559-652. http://physrev.physiology.org/content/93/2/599.long
Kulleperuma, K., S.M.E. Smith, D. Morgan, B. Musset, J. Holyoake, N. Chakrabarti, V.V. Cherny, T.E. DeCoursey, and Régis Pomès. (2013). “Construction and Validation of a Homology Model of the Human Voltage-Gated Proton Channel hHV1.” Journal of General Physiology. 141:445-465.
Musset, B., R.A. Clark, T.E. DeCoursey, G.L. Petheo, M. Geiszt, Y. Chen, J.E. Cornell, C.A. Eddy, R.G. Brzyski, and A. El Jamali. (2012). “NOX5 in Human Spermatozoa: Expression, Function and Regulation.” Journal of Biological Chemistry. 287:9376-9388.
Musset, B., V.V. Cherny, and T.E. DeCoursey. (2012). “Strong Glucose Dependence of Electron Current in Human Monocytes.” American Journal of Physiology: Cell Physiology. 302:C286-C295.
DeCoursey, T.E. (2012). “Voltage-Gated Proton Channels.” Comprehensive Physiology. 2:1355 1385.
Musset, B., S.M.E. Smith, S. Rajan, D. Morgan, V.V. Cherny, and T.E. DeCoursey. (2011). "Aspartate112 is the selectivity filter of the human voltage gated proton channel." Nature. 480:273-277.
Additional research
A more complete listing of DeCoursey’s published work can be found on PubMed.
Additional resources
- NSF summary of first dinoflagellate proton channel gene
- Wikipedia entry on voltage-gated proton channels
- Summary of recent Hv1 studies with special reference to phagocytes by Boris Musset (in German)
- A discussion of proton conduction by Antony Crofts
- Animations of the proton ATP synthase by Wolfgang Junge
- A useful nomogram showing the half-life of the superoxide anion before spontaneous disproportionation (courtesy of Ricardo Murphy)
- Chris Hartzell’s Chloride Channel Song
- A European view of American ethics on David Colquhoun’s website
Contact
Thomas E. DeCoursey, PhD
Professor
Rush University
Department of Physiology & Biophysics
Jelke Building
1750 W. Harrison St., Room 1245
Chicago, IL 60612
Phone: (312) 942-3267
Office phone: (312) 942-3684
Fax: (312) 942-8711